Pharma AI Visibility: Johnson & Johnson Serves Chrome 64/64, Blocks ChatGPT 0/64
Published 2026-04-20 · PROGEOLAB Research
The Pharma AI Visibility Index ranks the 14 pharmaceutical companies in the Fortune Global 500 on AI accessibility, llms.txt adoption, robots.txt AI policy, JSON-LD structured data, and WAF posture. Pharma sits at an unusual intersection: drug information is life-or-death content — prescribing information, contraindications, dosing guidelines, recall notices — that must be accurate and current. Yet the industry's regulatory caution around content distribution creates tension with AI-era information access.
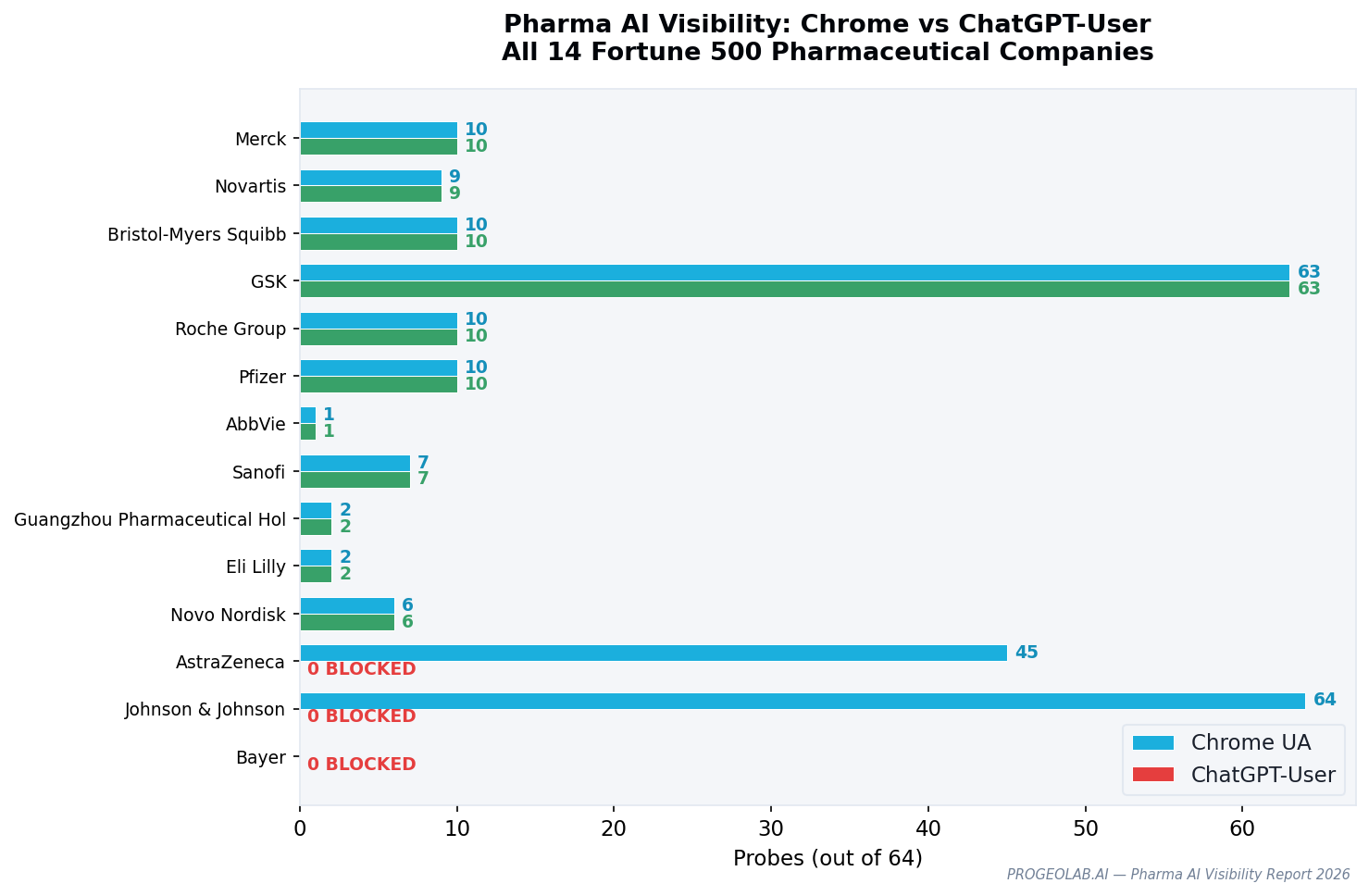
Pharma's gap rate is moderate: 2 of 14 companies block ChatGPT-User while serving Chrome browsers — a 14% rate, lower than telecom (27%) but above the Fortune 500 average of 10.6%. What makes the pharma finding distinctive is not the count but the cleanness of the block. Johnson & Johnson produces the single most precise example of AI-specific blocking in the entire dataset.
The J&J pattern: browser-OK, AI-blocked
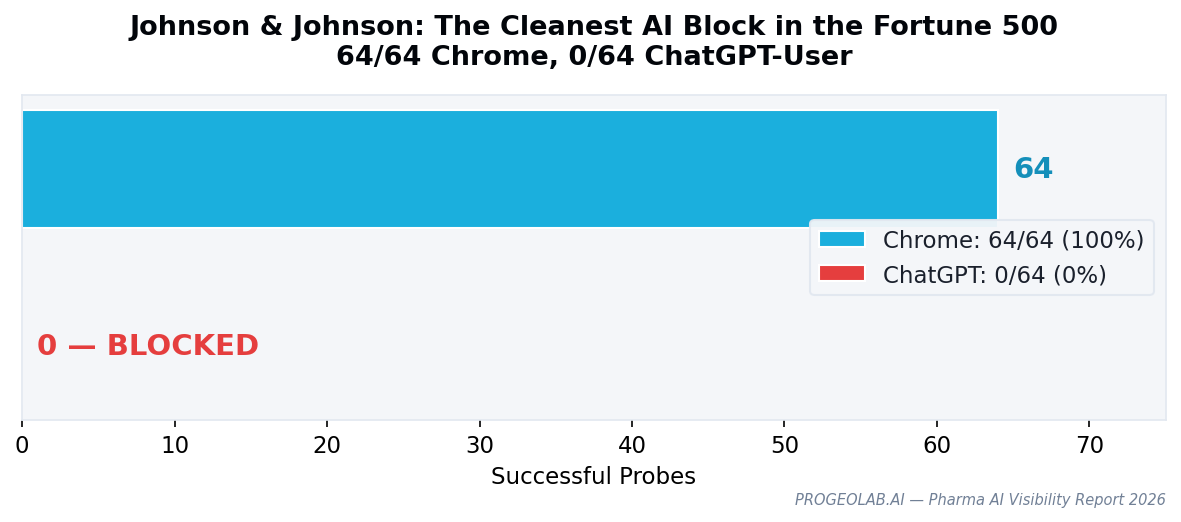
Johnson & Johnson's probe results across 64 endpoints:
| User Agent | Successful Probes | Rate |
|---|---|---|
| Chrome | 64 / 64 | 100% |
| ChatGPT-User | 0 / 64 | 0% |
Every single one of 64 HTTP endpoints returns content to a Chrome browser. Every single one blocks ChatGPT-User. This is not partial blocking or inconsistent WAF behavior — it is a comprehensive, deliberate policy of serving browsers while rejecting AI.
J&J has no JSON-LD on its homepage, no llms.txt, and no AI-specific robots.txt directives. The company has made no investment in AI discoverability and has actively invested in AI blocking. AI Readiness Score: 1.
The healthcare consequence: when a patient asks ChatGPT about Tylenol dosing, Motrin interactions, or Janssen immunology products, the model cannot access jnj.com. It answers from training data that may predate the latest FDA label update. J&J's own regulatory-reviewed content is replaced by whatever the model remembers — the AI Content Paradox in its sharpest form.
AstraZeneca: Googlebot trusted, ChatGPT rejected
AstraZeneca shows a different but equally instructive pattern. Chrome: 45/64. ChatGPT-User: 0/64. AstraZeneca is accessible to Chrome (45 of 64 endpoints) but blocks ChatGPT-User entirely. AstraZeneca also has JSON-LD on its homepage (Organization type) and is behind Cloudflare's WAF — suggesting sophisticated bot management is deliberately configured.
AstraZeneca's Cloudflare deployment likely uses Bot Management with AI-specific UA rules. The JSON-LD investment shows awareness of structured data for search engines; the AI blocking means that structured data is inaccessible to the AI crawlers most likely to surface it in answers.
GSK: the pharma leader
GSK operates at the opposite extreme. Chrome: 63/64. ChatGPT-User: 63/64. Nearly complete transparency to AI systems across more than 60 endpoints, plus JSON-LD structured data and a welcoming WAF posture. Among pharma companies, GSK is the most likely to be cited accurately in AI-generated health content — not because it has the strongest signals (Merck, Novartis, and Bristol-Myers Squibb match GSK's 5 score) but because GSK serves so much content so transparently.
When patients ask AI about asthma treatments, vaccine schedules, or oncology trials, GSK's current regulatory-reviewed content is reachable. When the same patients ask about J&J products, nothing current is available. The downstream narrative control asymmetry is real and measurable.
The Pharma AI Visibility Scorecard
| Rank | Company | Country | Chrome | ChatGPT | Score | Status |
|---|---|---|---|---|---|---|
| 1 | GSK | Britain | 63 | 63 | 5 | Accessible + JSON-LD |
| 2 | Merck | U.S. | 10 | 10 | 5 | Accessible + JSON-LD |
| 3 | Novartis | Switzerland | 9 | 9 | 5 | Accessible + JSON-LD, F5 BIG-IP |
| 4 | Bristol-Myers Squibb | U.S. | 10 | 10 | 5 | Accessible + JSON-LD, Imperva |
| 5 | Roche Group | Switzerland | 10 | 10 | 4 | Accessible, F5 BIG-IP |
| 6 | Pfizer | U.S. | 10 | 10 | 4 | Accessible, Cloudflare |
| 7 | Novo Nordisk | Denmark | 6 | 6 | 4 | Accessible |
| 8 | Sanofi | France | 7 | 7 | 4 | Accessible |
| 9 | Eli Lilly | U.S. | 2 | 2 | 4 | Accessible |
| 10 | AbbVie | U.S. | 1 | 1 | 4 | Accessible, Cloudflare |
| 13 | AstraZeneca | Britain | 45 | 0 | 2 | GEO gap, Cloudflare |
| 14 | Johnson & Johnson | U.S. | 64 | 0 | 1 | GEO gap |
The pharma distribution is narrower than automotive or banking: most companies cluster at 4-5 points, with J&J and AstraZeneca as clear laggards and nobody operating at the leading edge. No pharma company has an llms.txt. No pharma has AI-specific robots.txt directives. The sector treats AI accessibility as binary — either serve it (10 of 14) or block it (2 of 14) — with no company yet investing in curation for AI.
The compliance-visibility tradeoff
Pharma's regulatory framework (adverse event reporting, promotional speech restrictions, off-label use constraints) creates genuine reason for caution about content distribution. If AI redistributes drug information and users report side effects to ChatGPT rather than to the company's safety database, the adverse event reporting chain breaks. If AI summarizes prescribing information in a way that omits contraindications, the company's promotional speech obligations may be implicated.
These are real concerns. But J&J's blanket block does not solve them — it simply ensures that the AI content about J&J products is generated from whatever training-era information the model has, without benefit of J&J's current regulatory-reviewed content. GSK's approach — serve everything, let AI quote primary sources — may actually reduce adverse-event risk by replacing AI inference with AI retrieval of authoritative material.
What's in the full report
- Complete 14-company scorecard with Chrome, ChatGPT, JSON-LD, WAF, score per company
- J&J case study: the probe data, the likely WAF rule, and what patients see instead
- AstraZeneca case study: Cloudflare Bot Management configured against AI specifically
- GSK case study: what pharma-appropriate AI openness looks like operationally
- Regulatory context: FDA, EMA, and the adverse event reporting framework
- Compliance-visibility tradeoff framework for pharma legal and comms teams
- Recommendations for medical affairs, regulatory, and digital marketing organizations